DAT Practice Test (Survey of Natural Sciences)
Which organelle in a eukaryotic cell makes transport vesicles for exocytosis?
Correct answer: Golgi Body
The golgi body assists in the processing of proteins by modifying and sorting the protein. In addition, the golgi body is responsible for tagging and dispersing proteins that are destined to leave the cell. These proteins are packaged in transport vesicles that are then taken to the cell membrane, where the vesicle fuses with the membrane and ejects the protein outside of the cell. The ribosome and rough endoplasmic reticulum both help with protein synthesis but are not responsible for vesicle production. The mitochondria is the site of ATP production during cellular respiration and does not directly form vesicles. The smooth endoplasmic reticulum is essential in lipid production and detoxifying the cell.
The reaction quotient (Q) for a reaction involving nitrogen monoxide within a sealed flask was determined to be 1.1 × 10² at one point. Which reaction is most likely taking place in the flask if the amount of NO gas in the flask increased after this point?
Correct answer: N2(g) + O2(g) ↔ 2NO(g) Kc = 4.2 × 102
When Q > Kc, the numerator of the equilibrium expression (the product concentration) is too big, and the equation shifts to the left. This is true for both (D) and (B), meaning [NO] would decrease. When Q < Kc, the numerator/product concentrations need to increase. This is the case in (C) and (A), but NO(g) is only a product in (A).
When reacted with benzene, which of the following will produce an ortho/para configuration?
Correct answer: 2 of the below
Halogens, such as Cl- and R groups, such as CH3, both direct benzene in an ortho/para configuration. The other answer choices direct in a meta configuration. Know ortho/para/meta along with activating and deactivating groups from the heart!
In humans, cystic fibrosis is a recessive disease. What is the probability that a kid would be born with the condition if one parent is a carrier of the trait and the other parent is homozygous dominant, according to the principles of classical Mendelian genetics?
Correct answer: 0%
Diseases that are caused by recessive alleles require two copies of the allele. If a parent is homozygous dominant, he or she does not have any recessive alleles to pass to the offspring. The genotype for this parent can be represented by (CC). If the other parent is a carrier, he or she has only one copy of the recessive allele. The genotype for this parent can be represented by (Cc). This means that the offspring is guaranteed to received at least one dominant copy of the allele, and will have the genotype (CC) or (Cc). In both cases, the individual is not affected by the disease and the correct answer is zero percent (C). An affected individual's genotype would be (cc).
The cytosol's typical pH is at 7.2. The majority of enzymes in the lysosome, on the other hand, are only active at pH 5. This implies that
Correct answer: protons are actively pumped into the lysosome
The enzymes in the lysosome are enzymatically active because the pH inside the lysosome is 5. The pH is maintained by pumping H⁺ ions (protons) into the lysosome by active transport proton pumps into the lysosome membrane.
A compound is fully made up of silicon and oxygen atoms. What is the empirical formula of a compound containing 14.00 g of silicon and 32.0 g of oxygen?
Correct answer: SiO4
14.0 g of Si is 0.50 mol, and 32.0 g of oxygen is 2.0 mol. Converting that to a whole number ratio gives us 1 mol of Si for every 4 moles of O.
Which of the following functional groups has the highest priority in IUPAC nomenclature when numbering a parent chain?
Correct answer: Esters
Knowing how to name chemicals and nomenclature rules (including functional group priority) will give you the easy (and quick) points in this section.
Multiple binding sites exist on __________ enzymes.
Correct answer: Allosteric
All enzymes bind their substrates at their active sites. Enzymes that can be regulated by effectors (activators or inhibitors) that bind an additional site on the enzyme are referred to as allosteric enzymes. The correct answer is therefore (A). Competitive (D) and non-competitive (E) are terms used to describe inhibitory molecules that bind enzymes. All enzymes are catalytic (B), since they accelerate reactions, and all enzymes are regulatory (C) since they appear in pathways that control cellular processes.
Caffeine can attach to the adenosine receptor but cannot activate it since its structure is very similar to that of adenosine. When caffeine binds to the adenosine receptor, which of the following best characterizes the effect?
Correct answer: Caffeine blocks the effect of adenosine by preventing the binding of adenosine to the adenosine receptor
Caffeine is an adenosine signaling antagonist, blocking the effect of adenosine. When caffeine binds to the adenosine receptor, it blocks the G protein-coupled receptor from initiating the adenosine signaling pathway. As a result, PKA is not activated and drowsiness does not occur.
N₂(g) + O₂(g) + Cl₂(g) ↔ 2NOCl(g) ΔG° = 132.6 kJ/mol
What would happen to the value of ΔG° in the equilibrium below if the concentration of N2 were to increase, and why?
Correct answer: It would stay the same because the value of Keq would not change
Adding (or removing) any species in an equilibrium reaction does not change the equilibrium constant and also does not change the magnitude of the Gibbs free energy at standard conditions.
The melting point of which of the following is the highest?
Correct answer: 2,2-Dimethylpropane
The general trend is the higher # of carbons, the higher the boiling point and melting point. There is a specific rule for this however that involves symmetry and branching. If the compound has branching and symmetry, then it has a higher melting point than its non-branched and symmetrical counterpart, which, in this example, 2,2-dimethylpropane (having 5 carbons) and pentane (also having 5 carbons, but lacking any branches).
Involuntary muscles that do not contain striations are referred to as
Correct answer: smooth muscle
There are three types of muscles. Smooth muscles lack striations and are involuntarily controlled. Thus, the correct answer is (D). In contrast, skeletal muscles (C) are striated and are under voluntary control. Although cardiac muscle (E) is involuntary, it contains striations (E). Muscles are not characterized as autonomic, although autonomic means involuntary (A) and could refer to both cardiac and smooth muscles. There is no muscle tissue in the brain so choice (E) is not relevant.
Which of the following properties holds the water molecules in a vertical column of water in a plant's xylem vessels together?
Correct answer: the strong cohesion of property of water
Cohesion is the attraction between like substances. Because of the hydrogen bonding between them, water molecules stick to other water molecules, holding them together to form a vertical column in plant xylem vessels. The water molecules in the column also demonstrate adhesion, the attraction between unlike substances, when they form weak hydrogen bonds with the cellulose and other molecules in the walls of the plant vessels. This mechanism, however, helps the water column oppose the downward pull of gravity.
Three gases are contained in a sealed, rigid container: 28.0 g nitrogen, 40.0 g argon, and 36.0 g water vapour. What is the partial pressure of nitrogen if the total pressure exerted by the gases is 2.0 atm?
Correct answer: 0.50 atm
There is 1 mole of N₂, 1 mole of Ar, and 2 moles of water in the container. The mole fraction of nitrogen is 1/4 = 0.25.
PN₂ = (XN₂)(Ptotal)
PN₂ = (0.25)(2.0) = 0.50 atm
For this molecule, how many stereoisomers are there?
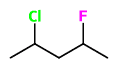
Correct answer: 4
The maximum number of stereoisomers is determined by the equation: 2ⁿ Where n = number of chiral centers. There are 2 chiral centers in the molecule above, thus rendering a maximum number of 4 stereoisomers.
In nucleic acids, which of the following is not a pyrimidine base?
Correct answer: Adenine
Nucleic acids contain bases that are classified as either purines or pyrimidines.
Purines in DNA and RNA include adenine (C), and guanine that is not listed.
Thyminc (D) and cytosine (A) are both pyrimidines in DNA, and uracil (B) is a
pyrimidine found in RNA. Therefore the correct answer is (C).
Bacteria that cannot survive in the presence of oxygen are known as:
Correct answer: Obligate Anaerobes
The term obligate means that this is a required condition that has to be met to ensure the viability of the organism. An obligate aerobe has to have oxygen present to be able to survive, like humans. Likewise, an organism that cannot live in the presence of oxygen would be an obligate anaerobe. Facultative anaerobes can survive in the presence of oxygen, but prefer aerobic environments. Permissive anaerobe is a term that does not exist in biology. Aerotolerant organisms do not require oxygen, but they are not poisoned by it either.
High-energy photons hit neutral chlorine atoms, forcing electrons to expel from the various filled subshells. After being ejected, which subshell's electrons would have the maximum velocity?
Correct answer: 3p
The farther away an electron is from the nucleus, the less ionization energy that is required to eject it, and as a result, the electron will have more kinetic energy after it is ejected. The 3p subshell is the farthest one that a neutral chlorine atom would have electrons in. Beware of choice (C); chlorine does not have a 3d subshell.
What is this atom's hybridization?
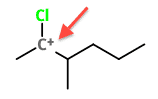
Correct answer: sp2
Be sure to pay special attention to the ‘+’ symbol indicating a cation. If it lacked a positive or negative sign, it would be a sp³ hybridization.
What is the empirical formula of an unknown compound with a carbon content of 48.02 percent, a hydrogen content of 18.74 percent, and a nitrogen content of 33.24 percent? (Nitrogen has an atomic weight of 14.0 g.)
Correct answer: C₅H₂₄N₃
The formula is C₅H₂₄N₃ and the correct response to the question is (C)- In choice (D), the subscripts are relative ratios of the percentages of each element in the compound. In choice (E), the moles of each compound was incorrectly calculated by place g/mol instead of moi/g. Choice (A) is a variant of (E), and choice (B) was randomly generated.
_________ is a type of species interaction in which one species benefits while the other is unaffected.
Correct answer: commensalism
The example to know is the cattle egrets that feast on insects aroused into flight by cattle grazing in the insects' habitat. The birds benefit because they get food, but the cattle do not appear to benefit at all.
SF₄(g) + H₂O(1) → SO₂(g) + 4HF(g) ΔH = -828 kJ/mol
Which of the following statements best captures the reaction described above?
Correct answer: The bond strength of the reactants exceeds that of the products
One way to calculate enthalpy is by subtracting the strength of the bonds broken in the reactants from the strength of the bonds formed in the products. If the first value is greater than the second value, the overall enthalpy change will be negative.
For the following reaction, which is a potential reagent for obtaining this product:
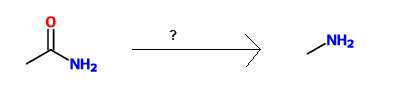
Correct answer: Br2, NaOH, H2O / heat
This is a Hofmann rearrangement reaction which converts a primary amide to a primary amine. Note the loss of the carbon.
12.5 g sucrose is dissolved in 0.100 kilogram water to make a solution. What is the solute's mass percentage in this solution?
Correct answer: 11.11%
The percent mass, or mass percentage of a solute in a solution, is the ratio of the mass of the solute to the total mass of the solution times 100. The mass of the solute is given as 12.5 g, and the mass of the solvent (water) is 0.100 kg. The mass of the water must be converted to grams, so 0.100 kg is the same as 100 g of water. Now the mass of the solute is divided by the total mass, so 12.5 g /(100 g + 12.5g) = 11.11 %, and the correct response is (E). Choice (C) is the value when 12.5 g of sucrose is divided by 100 g of water. This is incorrect because the denominator does not consider the total mass of the solution. Choice (A) is incorrectly calculated in the same manner as choice (C), without converting kilograms to grams. In choice (D) the mass of the solvent or 100 g is divided by the mass of sucrose or 12.5 g. In choice (B), the same calculation in choice (D) was used, however 0.100 kg is used instead of 100 g.
Inactive trypsin is a proteolytic enzyme secreted by the pancreas. The action of ________ can convert trypsinogen to trypsin.
Correct answer: enteropeptidase and trypsin
After releasing trypsinogen from the pancreas, enteropeptidase cleaves the inactive protein at specific sites. As a consequence of this action trypsin is generated. This enzyme has ability to generate trypsinogen on its own and to release an active enzyme.